What’s next for abortion pill after Supreme Court’s order

What’s next for abortion pill after Supreme Court’s order
Now that the high court has set out the rules that will govern access for now, the case can continue on its path through the courts.
Nothing will change for now. That’s what the Supreme Court said Friday evening about access to a widely used abortion pill.
A court case that began in Texas has sought to roll back Food and Drug Administration approval of the drug, mifepristone. Lower courts had said that women seeking the drug should face more restrictions on getting it while the case continues, but the Supreme Court disagreed.
The court’s action almost certainly will leave access to mifepristone unchanged at least into next year, as appeals play out, including a potential appeal to the high court.
The new abortion controversy comes less than a year after the Supreme Court’s conservative majority overturned Roe v. Wade and allowed more than a dozen states to effectively ban abortion outright.
The following is a look at the drug at issue in the new case, how the case got to the nation’s highest court and what’s next in the legal case.
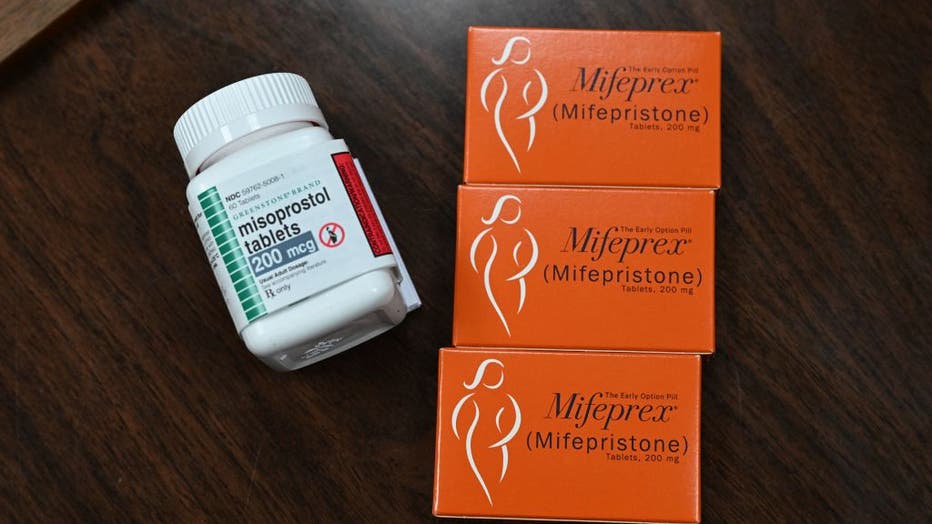
Mifepristone was approved for use by the Food and Drug Administration more than two decades ago. It has been used by more than 5 million women to safely end their pregnancies, and today more than half of women who end a pregnancy rely on the drug, the Justice Department said.
Over the years, the FDA has loosened restrictions on the drug’s use, extending from seven to 10 weeks of pregnancy when it can be used, reducing the dosage needed to safely end a pregnancy, eliminating the requirement to visit a doctor in person to get it and allowing pills to be obtained by mail. The FDA also approved a generic version of mifepristone that its manufacturer, Las Vegas-based GenBioPro, says makes up two-thirds of the domestic market.
What is Mifepristone?
(Photo by Robyn Beck / AFP) / RESTRICTED TO EDITORIAL USE (Photo by ROBYN BECK/AFP via Getty Images)
Mifepristone was approved for use by the Food and Drug Administration more than two decades ago. It has been used by more than 5 million women to safely end their pregnancies, and today more than half of women who end a pregnancy rely on the drug, the Justice Department said.
Over the years, the FDA has loosened restrictions on the drug’s use, extending from seven to 10 weeks of pregnancy when it can be used, reducing the dosage needed to safely end a pregnancy, eliminating the requirement to visit a doctor in person to get it and allowing pills to be obtained by mail. The FDA also approved a generic version of mifepristone that its manufacturer, Las Vegas-based GenBioPro, says makes up two-thirds of the domestic market.
Mifepristone is one of two pills used in medication abortions, along with misoprostol. Health care providers have said they could switch to misoprostol only if mifepristone is no longer available or is too hard to obtain. Misoprostol is somewhat less effective in ending pregnancies.
How did the case get started?
A lawsuit over mifepristone was filed in Amarillo, Texas, late last year. Alliance Defending Freedom, a conservative Christian legal group, represents the pill’s opponents, who say the FDA’s approval of mifepristone was flawed.
Why Amarillo? U.S. District Judge Matthew Kacsmaryk, who was nominated by then-President Donald Trump, is the sole district court judge there, ensuring that all cases filed in the west Texas city land in front of him. Since taking the bench, he has ruled against President Joe Biden’s administration on several other issues, including immigration and LGBTQ protections.
On April 7, Kacsmaryk issued a ruling that would completely revoke the FDA’s approval of mifepristone, but he put the decision on hold for a week to allow an appeal.
Complicating matters, however, on the same day Kacsmaryk issued his order, a court in Washington state issued a separate ruling in a lawsuit brought by liberal states seeking to preserve access to mifepristone. The Washington judge, Spokane-based Thomas O. Rice, whom then-President Barack Obama nominated, ordered the FDA not to do anything that might affect the availability of mifepristone in the suing states. The Biden administration had said it would be impossible to follow both judges’ directives at the same time.The Biden administration and the maker of mifepristone, New York-based Danco Laboratories, appealed to the Supreme Court, saying that allowing the appeals court’s restrictions to take effect would cause chaos. At first, facing a tight deadline, the Supreme Court gave itself some breathing room and issued an order suggesting it would act by Wednesday evening. But no decision came Wednesday and the court instead just gave itself an extension until just before midnight Friday. It wasn’t clear why.
The court did make its second self-imposed deadline, issuing its brief decision around 6:30 p.m. in Washington. Two conservative justices, Clarence Thomas and Samuel Alito, said they disagreed with the court’s action but no other justice commented.
What happens next?
The case is on a fast track. Now that the high court has set out the rules that will govern access for now, the case can continue on its path through the courts.
The 5th U.S. Circuit Court of Appeals has already announced it will hear arguments in the case in less than a month, on May 17. Both sides as well as interest groups will submit written briefs ahead of those arguments. And a three-judge panel of the court will hear the case, though the court has not yet said who those three judges will be. The group won’t issue a decision from the bench but instead hear arguments and ask questions. That will give the public a sense of what they’re thinking. Their decision will be made privately after oral arguments, and at some point they’ll issue a written decision announcing it.
Both sides then have an opportunity to appeal, taking the case to all the judges of the appeals court or directly to the Supreme Court. The justices take a break for the summer, however, and don’t start hearing cases again until October.

